Categories
- Pipe & Tube (18)
- Flange & Fitting (97)
- Fastener & Gasket (12)
- Valve & Pump (18)
- Base Material (11)
- Equipment (8)
- Application (31)
- Technical (110)
Austenitic stainless steels, on exposure to temperatures in some certain range for a time long enough, become susceptible to intergranular corrosion. This susceptibility has been termed “sensitization”, which means that the steel is “sensitized”. Sensitization of austenitic stainless steels has been attributed to the precipitation of M23C6 carbides on the austenite grain boundaries. The development of scanning transmission electron microscopes (STEM) with electron beam sizes of about 10 nm in diameter, coupled with energy-dispersive x-ray analysis, has provided direct proof of this theory.
The mechanism for sensitization is that time at certain temperature will determine the amount of carbide precipitation. When the chromium carbides precipitate in grain boundaries, the area immediately adjacent is depleted of chromium. These areas are impoverished in chromium and are termed depleted zones. The STEM studies have proven that increasing holding time caused the width of the depleted zone to increase. The width of chromium depletion along the grain boundaries around individual carbides was much greater than the width of the depletion zone into the grain interior. Since the passivation of chromium plays a main role in corrosion resistance, the depletion of chromium along grain boundaries renders the austenitic stainless steel much more susceptible to intergranular corrosion.
Except for intergranular corrosion, sensitization also lowers resistance to other forms of corrosion, such as pitting, crevice corrosion, and stress corrosion cracking (SCC). Care must be taken when austenitic stainless steels are subjected to certain welding or service conditions. Long time exposure to the sensitization temperature shall be averted.
Three factors, time, temperature, and chemical composition, in combination determines the sensitization of the austenitic stainless steel. Take the most common and basic austenitic stainless steel type 304 for example:
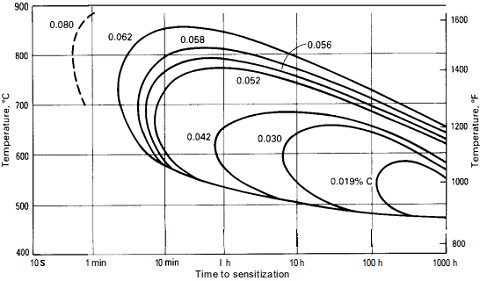
Figure-1: the time-temperature sensitization curves for TP304 with various carbon contents.
Figure-1 has illustrated the time-temperature sensitization curves for ss 304 with various carbon contents. A type 304 stainless steel with 0.062% carbon would have to cool below 595°C [1100°F] within about 5 minutes to avoid sensitization, but a type 304L with 0.030% carbon could take about 20 hours to cool below 480°C [900°F] without becoming sensitized. The lower the carbon content, the better its resistance to sensitization. Complete immunity requires a carbon content between 0.015% to 0.020%.
Additions of titanium and/or niobium can also tie up the carbon effectively preventing sensitization as long as the ratio of these elements to the carbon content is high enough. Since these elements have an affinity for carbon and form carbides easily; this allows the chromium to remain in solution even for extremely long exposures to temperatures in the sensitizing range. The addition of titanium and/or niobium is termed stabilization. Stabilized austenitic stainless steels, namely types 321, 347, 348 and UNS N08020, contain controlled amounts of titanium or of niobium, which render the steel nearly immune to intergranular precipitation of chromium carbide and its adverse effects on corrosion resistance. Nevertheless, these alloys may require annealing to relieve stresses, to increase softness and ductility, or to provide additional stabilization.
Due to the diverse chemical compositions (especially the carbon, titanium & niobium contents), the sensitizing temperature range for each austenitic stainless steel is quite different. For example, there are various technical literature stating sensitizing ranges of types 304 as 425°C~870°C, 480°C~815°C, and 540°C~845°C, etc. The holding time at temperature is also very important. Only continuous soaking in the sensitizing temperature can lead to sensitization. If the holding time is very short, there will be little chromium carbide precipitation hence no significant deletion of chromium and sensitization will not happen. This leads to the only way to correct sensitization of austenitic stainless steel: solution annealing.
